
Publisher:
Bonnie King
CONTACT:
Newsroom@Salem-news.com
Advertising:
Adsales@Salem-news.com

~Truth~
~Justice~
~Peace~
TJP
Feb-27-2010 02:47

 TweetFollow @OregonNews
TweetFollow @OregonNews
John Stossel and Truth, Justice and the American Way
Marianne Skolek Salem-News.comStossel buys the marketing ploy of a shrewd criminally convicted pharmaceutical company.
 John Stossel appearing on a Fox News program. Baby with Thalidomide based birth defect courtesy: Wikipedia |
(MYRTLE BEACH, S.C.) - This week John Stossel of ABC-TV 20/20 fame, a consumer reporter, investigative journalist, libertarian columnist and Fox Network journalist, posted the following information as a plug for his new television show:
"People who are desperate to try different medicines and treatments run into a wall of FDA bureaucracy. People suffer and die because the government 'protects' us. It should protect us less and respect our liberty more."
 Marianne Skolek |
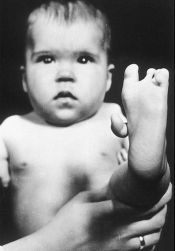
The libertarian Stossel further asks -- Why, in our "free" country, do Americans meekly stand aside and let the state limit our choices, even when we are dying? This week’s show includes Stossel's confrontation with Terry Toigo of the FDA. She calmly explained that such restrictions are necessary to protect the integrity of the government’s safety review process -- until Stossel shouted: "Why are you even involved? Let people try things!" Toigo replied, "We don’t think that’s the best system for patients, to enable people to just take whatever they want with little information available about a drug." Fortunately for us, Stossel wasn't defending the use of the drug Thalidomide, used by pregnant women who were experiencing sleep problems back in the early 1960's. Thalidomide was sold in a number of countries across the world from 1957 until 1961 when it was withdrawn from the market after being found to be a cause of birth defects in what has been called "one of the biggest medical tragedies of modern times". The Thalidomide tragedy led to much stricter testing being required for drugs and pesticides before they can be licensed. Maybe Stossel should go into the FDA website and read up on something under the heading - Can any individual patient qualify for expanded access to any drug? It reads as follows: FDA regulations allow access to investigational drugs for treatment purposes on a case-by-case basis for an individual patient, or for intermediate-size groups of patients with similar treatment needs who otherwise do not qualify to participate in a clinical trial. They also permit expanded access for large groups of patients who do not have other treatment options available, once more is known about the safety and potential effectiveness of a drug from ongoing or completed clinical trials.
Just as in clinical trials, these investigational drugs have not yet been approved by the FDA as safe and effective. They may be effective in the treatment of a condition, or they may not. They also may have unexpected serious side effects. It is important for you to consider the possible risks if you are interested in seeking access to an investigational drug.
In order for a patient to gain access to an investigational drug outside of a clinical trial, the patient must have a serious or immediately life-threatening disease or condition and no comparable or satisfactory therapeutic alternatives. Additionally, the drug manufacturer and the patient’s doctor must make special arrangements to obtain the drug for the patient. These arrangements must be authorized by the FDA. These safeguards are in place to avoid exposing patients to unnecessary risks. Manufacturers may not always be willing or able to provide access to a drug outside of their clinical trials. Physicians may not always be able to seek expanded access for patients, depending on a patient’s medical history and the risks associated with taking an investigational drug. The physician must determine that the probable risk from the drug is not greater than the probable risk from the disease. Not all physicians are willing to manage the use of an investigational drug for patients in their care.
 Although arrangements for these investigative drugs must be authorized by the FDA, the "manufacturer and patient's doctor must make special arrangements to obtain the drug for the patient." I wonder how many drug manufacturers and doctors are willing to risk the liability of exposing patients to a drug unapproved for use by the FDA? You might want to consider that Mr. Stossel.
And then Stossel attacks the Drug Enforcement Agency because as he says, they "watch pain-management doctors like hawks. Drugs like Vicodin and OxyContin provide wonderful pain relief. But because they are also taken by 'recreational' drug users, doctors go to jail for prescribing quantities that the DEA considers 'inappropriate.' As a result, pain specialists are scared into underprescribing painkillers. Sick people suffer horrible pain needlessly."
Although arrangements for these investigative drugs must be authorized by the FDA, the "manufacturer and patient's doctor must make special arrangements to obtain the drug for the patient." I wonder how many drug manufacturers and doctors are willing to risk the liability of exposing patients to a drug unapproved for use by the FDA? You might want to consider that Mr. Stossel.
And then Stossel attacks the Drug Enforcement Agency because as he says, they "watch pain-management doctors like hawks. Drugs like Vicodin and OxyContin provide wonderful pain relief. But because they are also taken by 'recreational' drug users, doctors go to jail for prescribing quantities that the DEA considers 'inappropriate.' As a result, pain specialists are scared into underprescribing painkillers. Sick people suffer horrible pain needlessly."
Look up the meaning of the words "pill mills" Mr. Stossel and look into the doctors the DEA "watch like hawks" before you spew the nonsense you have been hand fed by pain societies in existence because the maker of OxyContin, Purdue Pharma keeps them financially fat. Stossel further writes that "The DEA told us that good doctors have nothing to worry about. But Siobhan Reynolds, who started the Pain Relief Network after her late husband was unable to get sufficient pain medicine, says the DEA’s cherry-picked medical experts persuade juries that they should jail any doctor who administers higher doses of pain relief than the DEA’s zealots think appropriate. News of those jail terms spreads. Doctors learn to be stingy with pain meds."

No, Mr. Stossel doctors go to jail because patients are dying under their care and in large numbers. Look at the "pill mills" or pain clinics who have lines of people waiting to have their OxyContin prescriptions filled by physicians who do not follow any standard of care for their patients. Ask the DEA how many patients have died as a result of these physicians being charged for their over zealous prescribing of OxyContin. No one should suffer pain needlessly in this country and no one should die in a motel room when any emergency room will treat any patient with a physical condition needing proper medical attention. In case you're not aware, Mr. Stossel, scores of parents all over the country are dealing with the death and addiction of their children because of the criminal "overmarketing" of OxyContin and not the "undertreatment of pain" marketing ploy you bought into by a very shrewd criminally convicted pharmaceutical company and their heavily funded pain societies. I wonder what Mr. Stossel's next investigative reporting will cover in order to protect us less and respect our liberty more. How about the elimination of speed limit signs -- after all we're sick of being protected. Somehow I hear Perry White shouting "Don't call me chief" -- but it is John Stossel, cub reporter being yelled at -- and not Jimmy Olsen.
=========================================== Salem-News.com Reporter Marianne Skolek, is an Activist for Victims of OxyContin throughout the United States and Canada. In July 2007, she testified against Purdue Pharma in Federal Court in Virginia at the sentencing of their three CEO's who pled guilty to charges of marketing OxyContin as less likely to be addictive or abused to physicians and patients. She also testified against Purdue Pharma at a Judiciary Hearing of the U.S. Senate in July 2007. Marianne works with government agencies and private attorneys in having a voice for her daughter Jill, who died in 2002 after being prescribed OxyContin, as well as the voice for scores of victims of OxyContin. She has been involved in her work for the past 7-1/2 years and is currently working on a book that exposes Purdue Pharma for their continued criminal marketing of OxyContin.
Salem-News.com Reporter Marianne Skolek, is an Activist for Victims of OxyContin throughout the United States and Canada. In July 2007, she testified against Purdue Pharma in Federal Court in Virginia at the sentencing of their three CEO's who pled guilty to charges of marketing OxyContin as less likely to be addictive or abused to physicians and patients. She also testified against Purdue Pharma at a Judiciary Hearing of the U.S. Senate in July 2007. Marianne works with government agencies and private attorneys in having a voice for her daughter Jill, who died in 2002 after being prescribed OxyContin, as well as the voice for scores of victims of OxyContin. She has been involved in her work for the past 7-1/2 years and is currently working on a book that exposes Purdue Pharma for their continued criminal marketing of OxyContin.
Marianne is a nurse having graduated in 1991 as president of her graduating class. She also has a Paralegal certification. Marianne served on a Community Service Board for the Courier News, a Gannet newspaper in NJ writing articles predominantly regarding AIDS patients and their emotional issues. She was awarded a Community Service Award in 1993 by the Hunterdon County, NJ HIV/AIDS Task Force in recognition of and appreciation for the donated time, energy and love in facilitating a Support Group for persons with HIV/AIDS.
oxydeaths.com/news_chilling.htm
nytimes.com/2007/05/10/business/11drug-web.html?
blog.nj.com/njv_bob_braun/2007/07/sometimes_only_justice_can_rel.html
judiciary.senate.gov/hearings/testimony.cfm?id=2905&wit_id=6612
You can send Marianne an email at: mskolek@aol.com
oxydeaths.com
Articles for February 26, 2010 | Articles for February 27, 2010 | Articles for February 28, 2010




Salem-News.com:
Terms of Service | Privacy Policy
All comments and messages are approved by people and self promotional links or unacceptable comments are denied.
Amanda March 17, 2012 1:55 am (Pacific time)
OxyContin should be taken off the market. I have known people over the past decade acuiring the pills for recreational use. They enjoy the High it produces. After long-term use, coming off the Drug is very difficult and often those people need to go into a Methadone clinic for further treatment. Another example of medication that was benefical but taken off the market because of abuse is Quaalude. I think OxyContin should go up for review, and acknowledge it is abused more often than it's originally prescribed reason, for serious pain. Please don't let mega profits outweigh the harm it does to both children and adults.
Henry Massingale October 25, 2010 11:13 am (Pacific time)
It has hit the streets and now people are asking whats up with this International Boycott Of The Arabic Drug Empire ? Why so many supporters and no one is challenging this movement. We vote no to Prop: 19 Welcome to the , International Boycott Of The Arabic Drug Empire Phase 3 I can show you why not, this is a insult to the Men and Women of Police Enforcement who died fighting in this War Against Drugs. I can just see it now A Government Run Drug Empire. This was tried with heroin, and over 100,000 Americans Died from Pharmaceutical Heroin, information was released by FDA. Now a ugly truth that at first I thought to be a lie,Our Constitutional Laws are violated over this War and the Issues within. A lie and a deal to sale heroin In the USA. American Military Personal dieing in the poppy fields to protect the poppy plant from their original owners. A betraying of the American People to the deal made with Ben Laden and his Drug Lords. What bleeeppps me off the worse, I wanted one person to say this was in error, of what I see and what I found in my little investigations, by adding up 2+2......... Welcome to, International Boycott Of The Arabic Drug Empire Phase 3 just look it up on the net, and see thousands of post and supporters..... Henry Massingale / FASC Concepts in and for Pay It Forward covers the web post on google Drop by and see why we built a anti crime / war form in a Health Care Reform Concept. To strategically Rebuild America www.fascmovement.mysite.com on google look for page 1 american dream official site
Henry Massingale August 17, 2010 9:55 am (Pacific time)
Todays Health Care Programs is a complete failure because of the concepts of heroin made pills under a Health Care Concept and the FDA reported around 100,000 Americans dead. Now this Heroin has brought the United States into a War over drugs. “Post 9/11”... Lets just say all of this is true, and supported with News Reports by ABC News, CBS News, NBC News, and Official FBI investigations. I did note after 9/11 that the failures of of Home Banking Financing, And the cost of these failures was at the cost to Tax Payers. The crash of the Stock Market at $980 Billion dollars in one day disappeared and I do not remember the firm that lost the most invested dollars, but I think it was the AARP. At any rate this takes me right back to the 2 different attacks against the World Trade Center. Why this one Building ? Over a year ago I asked , why is there a hands off policy that prevents the destruction of the poppy fields. One year later I was still asking why ? I had no idea that Heroin was being sold in the USA as health care pills, for pain..... My whole concept with my Boycott Of The Arabic Drug Empire, went south and north and to what is now called the source. The Purdue Pharmaceutical Company, so please note this.. The Purdue Pharmaceutical Company, invested Billions of dollars into different companies through different sources, in order to cover the billions per month earned from selling heroin addiction. This is called a paper trail. These, hundreds of investments were done to launder this money in case of a addiction epidemic takes place in the United States. Now I am willing to bet, a name, a company name through this paper trail will lead to other insider trading and making money from the death of the People on 9/11. A lot of people handle this information carefully because they fear a link to Organizes Crime Figures in America. I assure you this, I am after one company that failed these Crime Figures, I am after the poppy fields and to end this Damn War Based on a lie, in part, of why we are at War. These Organized Crime Figures, from the Streets to the Families, do not want any part of this because it is a insult to the whole world of Man, and this Attack was a statement to pin point, of being betrayed within a deal made to sell Heroin. You do not make deals with the Family or Drug Lords and think because you may have the support of a Official of Government to back you up that you are safe. I will tell you all this one more time, I am dyslectic and because of this curse, I can see into the truth and lies of issues and People. I stay far from the connection of Man because of this curse, and I have never opened my mouth for over 30 to 40 years. Now I am in this Matrix, and I allow my mind to see that of which I do not wish to see. The Matrix of the Internet is buzzing as this truth unfolds before all of the People of the United States Of America. And All I wish for is to go back to work as a painter, and be within the hills of Tennessee, where I belong. My whole web site was built for President Obama, my love for all my children, my faith in God and Country. My mind is tired of seeing this world of betrayal because of a Dollar. And I am one of few to betray my Faith, I will use this Faith and this War has ended and the connection that all men and women sense to a past that is ancient in origin flows through this world as we look at the sky or a tree. My task is all most completed.... Is the Officials of the United States Government willing to kill to control the Heroin Trade in Afghan, because of this dollar ? Henry Massingale / FASC Concepts in and for Pay It Forward covers the web post on google Drop by and see why we built a anti crime / war form in a Health Care Reform Concept. To strategically Rebuild America www.fascmovement.mysite.com on google look for page 1 american dream official site
byMassingale July 21, 2010 8:58 am (Pacific time)
As it would seem, the Social Grace of the Rich is above the Law as long as the dollar support is at the back door. Do you wish to end this War and this Drug Epidemic , then stage a protest march that will awaken the American People, then take this War / drug issue against Purdue Pharmaceutical Company.... I am tired of seeing our Men and Women of the United States Military die as the American Dollar supports our enemy. Join Us...in the International Boycott Of The Arabic Drug Empire Phase 2 Welcome to FASC Concepts , The Day The World Stood Still , We are at War with a Drug Empire For nine years we have been at war and I still ask the big why ? Why is there a hands off policy that prevent the destruction of these poppy plants ? Did you know that American Dollars supported the Terrorist Attack on 9/11/2001, and thousands of lives have been lost. Who are the investors of the Pharmaceutical Company Purdue that makes Oxycontin and how are they above the Law ? Oxycontin is Heroin made from the opium / poppy plants. It is Illegal by Law to buy and resale Heroin. Purdue is responsible for the deaths on 9/11 and the on going addiction here in America. If it was Purdue who wished to be known as the largest heroin dealer in the USA, well congratulations and for showing the people who you do business with. Purdue, you guys just dont get it, you all look like a bunch of idiots in the face of our enemy, and the American People look stupid. You all at Purdue are now in my way. This drug from the poppy plant and the fight against its use goes back around 4000 to 6000 years and man seeks to control that which has shown that the poppy plant is as nature selected, uncontrollable. This is like a old saying as the Scorpion stung the Frog as he swim across the water and the Frog said why did you do this , now both of us will die ? And the Scorpion said it is my nature.... This is my Little War against the Arabic Drug Empire. Please allow me to share a concept with the People Of America, that will build for tomorrow. The Great Wall Of China in America. A 10 year project, and within the respect of why China built that wall and even as of today China makes money from that wall. This project will build towns and cities on both sides of the border and will bring peace and security. Another concepts is, To find a way into the hearts of children, first you must allow a truth to be shared and then let this share build within their hearts and you will see children open their minds to things not known. I read these things and I see where the Arabic Drug Empire is involved with the Mexican Drug Lords. What lost of understanding is that according to the faith of Bin Laden it is forbidden by God to walk among the infidels. That all infidels are to be vanquished from this world in order that the chosen ones will repopulate the world. It is a well known fact that the Arabic Drug Empire seek biological war tactics and it has been heard that a chemical balance is sought of how to implement Biological with Drugs, in order for it to be undetectable. The betraying of the Mexican Drug Lords by The Arabic Drug Empire will bring death to Mexico and the United States. Why should the Mexican Drug Empire take faith in my words, first If you and I stood before God I say to you that this is true it is the words of the streets that hold credit, and nothing is offered to them, The Mexican Drug Lords, dealing drugs is illegal. I know that some of the issues I write about is a bit over the top, but hey, if this post, by me makes you mad, welcome to a mad, mad drug world. I wonder how much in tax dollars this is going to cost the American People..... So join Us International Boycott Of The Arabic Drug Empire Phase 2 Henry Massingale / FASC Concepts in and for Pay It Forward covers the web post on google Drop by and see why we built a anti crime / war form in a Health Care Reform Concept. To strategically Rebuild America www.fascmovement.mysite.com on google look for page 1 american dream official site
Lyn March 1, 2010 6:32 am (Pacific time)
If Purdue Pharma had never "mis-branded" Oxycontin and over-marketed it to the extent that they did, then physicians would not be fearful of prescribing opioids like they are today. Prior to Oxycontin hitting the market, doctors were reluctant to prescribe appropriate amounts of opioids to adequately treat pain. Purdue was well aware of that fact and took advantage of it when they blatantly over-marketed Oxycontin. If you were to examine the number of doctors who have ever been arrested for over-prescribing opioids, I would bet a great deal of money that most of those convicted took place AFTER Oxycontin hit the market and Purdue convinced doctors to over-prescribe Oxycontin. Thanks so much, Purdue, for making legitimate physicians treating legitimte pain patiennts MORE reluctant to appropriately treat pain. Oh, and thanks to Purdue for making so much money from the sales of Oxycontin so that their owners are now extremely wealthy, as is anyone who works for the company during the Oxycontin timeframe.
Mills February 28, 2010 2:00 pm (Pacific time)
I am so thankful that we live in a country where we have a free flow of information from so many different sources to help us make decisions on the myriad of things that impact our lives, whether it be critiques on medicines, television programming or other news sources we have available. Obviously that standard saying "Different strokes for different folks," is what gives the free market system it's zest. Can you imagine living in one of these countries where a cabal tells you what you can read or watch because they know better than you? The American consumer votes not only with a ballot, but also with their time and money. If you don't have what they want, then woe to you. Trends always change, as does customer loyalty, suffice what is popular now most likely will not be so in the future. I think the exception to that is in the area of news reporting, if you keep it true, your audience will stay. Americans can sniff out the truth, eventually, regardless who tells them something is otherwise.
Hank Ruark February 28, 2010 10:24 am (Pacific time)
C.B: Fox is cunningly, cannilly and knowingly constructed to call forth precisely the level and depth of your response. That's both the danger and the desperation attachable now to its impact on far too many unaware viewers immersed in its massive misdirections, distortions and too-often plain perversions of events, facts and fancies of those who pay the bills, purchase the one-time professionals now in thrall to their current location, and control the content and presentation. It was constructed by Ailes to Murdoch design, paid for by Murdoch dollars, with purpose and programming built to his marque for massive propaganda purposes --at which it is only partially successful since not all who view it fall for its too-facile perpetrations. Have you surveyed the three major journalism-field reviews on this, sir ? Have you read the Murdoch and Ailes biographies and any similar reports of their professional activities ? Mine reports professional assessments within the news industry,by those particularly and peculiarly trained by both special experience and their own academic preparation for that evaluation -- including those references just given. What's your level in those very relevant areas,sir --and your own practical working experience, both personnally and in-family ? My four sons have all worked at least 20 years each in the industry, two now still active have more than 40 years each, still hacking...and mine own time includes 50 years split between education and these fields. It was from allathat that I shared assessment --not only and merely from personal feeling built on the usual casual viewing you seem here to project. IF you have links to other professional reports from any reliable source, please now send those links so we can now "see with own eyes --evaluate with own mind", our ongoing pattern in this open, honest, democratic S-N channel. Dissent is essential to the conversation at the heart of democracy --but it must be the informed and factually-founded kind and level. So you pays your nickel and then your attention to one of us...
Steve Walker February 28, 2010 6:14 am (Pacific time)
On Oxycontin - I do know about it. My wife needed pain killers for her surgeries and for the pain caused by the disease near the end. One of them was Oxycontin. It was a very helpful drug for her, but it does not cause me to think or believe that it is the right drug for everyone, or that there shouldn't be effective controls on its prescribing and use. As my life came back together years after my wife's passing, I met a nurse who works in hospice (no connection to my past experiences) and we are now together. We talk extensively about thorny issues like these, and you might be surprised to find out how medically necessary it is to have multiple options for effective pain control when dealing with very sick people. Yes, I know about Oxycontin and opioids and other forms of pain medications. I have taken some myself for short periods under the care of a doctor for while recovering from orthopedic injuries and surgeries. Another thing I have learned over the years, and for which I have been sometimes sternly rebuked (especially at the start when I was very angry about had happened to my wife) is that it is wrong to assume people who disagree with us don't understand something important. I have since come to learn that I learn the most from people who disagree with me. The problems we have are often caused by people who don't do that. Nothing fails faster and ore consistently than people making decisions in an echo chamber. Not being argumentative here, just making a point that has made me a better advocate.
Steve Walker February 28, 2010 6:03 am (Pacific time)
All good comments. I actually think Marianne and I have a lot in common. My wife of 20 years was denied her chance to survive a terminal form of cancer by an FDA more focused on its internal processes than on advancing the public health, then denied multiple times a clear path to extending her life. We sought access to investigational drugs that obviously worked for some patients. Every one of them was eventually approved for her form of cancer, but too late for her to get them when they might have saved her life. I think we have both learned how difficult and frustrating it is to get bureaucrats and politicians to first listen, and then to act. We at the Abigail Alliance learned a long time ago that we don't need any enemies, and because of what happened to us (and continues to happen to tens of thousands of people every year), we need to do a lot of listening to fully understand how to get others to agree with us and to help us find solutions - because the people who disagree with us are almost always doing so for a reason that turns out to be important. They just have a different perspective because they were tragically or positively affected in a different way. To correct the record, I am not an expert on Stossel, I am an expert on the problems with FDA's drug approval and development process - which often impacts directly on people with serious and terminal diseases in very adverse and even lethal ways. All deaths are a tragedy, and they are made even more tragic when the people responsible for preventing them fail and contribute to or even cause those deaths. It is sometimes difficult to understand the multiple sides of issues as complex as whether certain medications should or should not be available, to whom they should be available, when they should be available, how they should be controlled, etc., and our system does not always do a very good job at this. On that, I hope we can all agree. It is, unfortunately, the responsibility of people negatively affected by the mistakes to try to fix the problems that lead to them being made in the first place. I know a lot about the Anna Tomalis story, the young lady who struggled with and died from a rare form of sarcoma that was highlightred on the show. It really happened, it really was tragic, it was a major failure of our system, and it is not at all a rare occurrence. It happens a lot. The way in which she and her family were treated by the FDA was outrageous, and I give Stossel credit for recognizing that we have problems at the FDA that many others don't know about. He was clear at the end of his show that his proposed solution is his opinion. What we at the Abigail Alliance think should be done about the access issue is posted on our website.
P.N. Hotra February 27, 2010 10:34 pm (Pacific time)
It's a shame that we tend to demonize a medication, due to a few bad apples. It is this very over-reaction that keep "real" doctors from entering into the field of pain management. If anybody cares, clinical studies shows that indeed pain is undertreated at all levels in this country and around the world due to the misinformation that the treatment of pain with opioids will inherently lead to addiction and abuse, when in fact the opposite is true. Whether you're dealing with morphine or oxycontin or any other powerful pain killer, the danger is the same when used incorrectly, or presribed incorrectly and the intentions of everyone involved. I know how devastating loosing a loved one to an addiction, an adverse drug interaction and medical mistakes can be. And yes, some of the problem is due to a very few unscrupulous doctors and those in the illegal drug trade that get these among all the other drugs in the black market illegally. Medical decisions should be made between the doctor and their patients. And doctors should not be scared to treat their patients for any condition. Don't forget, every new barrier for an already beaten down chronically ill patient adds to the burden of an overly burdened population. These medications can be a godsend for some. Every year millions of Americans live and die with under treated pain. Even those in hospice. So please, we need to be constructive in this most personal of matters and try not to make any knee-jerk reactions. Godbless.
Conrad Byrnes February 27, 2010 6:28 pm (Pacific time)
The FOX Network provides a cross-section of opinions and allows all views to be expressed in a professional manner during their news programming. They have people of different political parties on every day sharing their perspectives. Recently the First Lady was on Govenor Huckabee's program, should we not hear her topical views because of who owns FOX? If we take and expand degrees of owner/CEO's relationships/connections to news departments and/or entertainment programming, then I guess for some no programming is fit to watch unless it fits their values. I say that as a viewer and a voter, aka, an American citizen who is free to watch and read whatever I want. I often turn off the television or change channels during some programs, like O'Reilly and others of similar blowhard opinions.
Hank Ruark February 27, 2010 2:56 pm (Pacific time)
Steve W. et al: You say you're expert on Stossel, sir, but what you offer is expertise and public service on his limited role re drug use, as outlined in depth here by Marianne S. Happens I am something of an expert (perhaps equal to your specialty ?) re both Stossel as libertarian and Fox News in the role custombuilt for them by Roger Ailes and the Murdoch dollars. All you need know re Stossel is his libertarian stance, laid out in depth by M.S. All you need know re Fox News is that it was built to order by Ailes for Murdoch. Knowledge in those two areas kills any conceivable reliance on either Stossel or Fox News, for any responsible person with a penchant for even the most penurious of cogitation.
Pete Jackson February 27, 2010 2:22 pm (Pacific time)
I too have never made a dime Mr. Walker, and I too am expert at one aspect of this issue, you tend to become one when your daughter is killed by one pill of OxyContin. The current medical literature is full of research confirming the addictive qualities - to patients as well as nonpatients - and other adverse side effects of the most potent opioids, the extended-release opioids like OxyContin, which was never clinically proven to be any more effective than the lower dose opioids that were already on the market. I don't imagine you were aware of that, were you?
Johnny February 27, 2010 11:29 am (Pacific time)
so what should doctors prescribe for victims of a drunk driver who are in need of pain relief? If such victims are permanently disabled due to severe spinal pain do we say to them that they should not get heroin-like substances due to the chance they might become addicted?
Steven Walker February 27, 2010 6:17 am (Pacific time)
(Typos corrected) I am something of an expert on the subject of John Stossel's show on access to medical breakthroughs, and much of what he said about the FDA being a barrier to the delivery of proven progress to dying patients is true. The FDA's expanded access programs actually barely work, and are unavailable to most patients. If you want to know more about this, then visit www.abigail-alliance.org. I have been directly involved for about ten years in trying to improve those programs and make them more accessible for patients who have serious and life-threatening diseases, like for example terminal cancer, and can't get into a clinical trial (which is most of them). While it is certainly reasonable to disagree with Mr. Stossel's proposed solutions, if you are going to opine on something like access to investigational drugs for dying patients with no other options, you should first learn the facts. The FDA really is a big problem in this area, and the reasons for that would surprise most Americans. I actually was pleased that Mr. Stossel got his facts right with regard to FDA's handling of access to investigational drugs, even if most would disagree that getting rid of the FDA altogether isn't the answer. As for the powerful painkillers he discussed, for patients who need them, they really are miracle drugs. The problem isn't that we have effective pain medications. It is the mishandling and misuse of those drugs by a small number of persons, and what has become an over-reaction to the problem by the DEA. Many patients who have real, severe pain, for example people in nursing homes, are now routinely undermedicated because of this over-reaction. There are always two sides to complicated stories like this, and Stossel was trying to tell the side that doesn't get much press. An informed opinion requires in depth knowledge of the facts and all sides of an issue from the perspective of people affected by it. If you take a can objective look at the side Stossel was explaining, you will find it is real, and is not being advanced by drug companies. It is being advanced by people like me, a volunteer patient advocate who has never made a dime from it.
Ada February 27, 2010 6:01 am (Pacific time)
Thank you Marianne for bringing the real issues to light. It is obvious that Mr. Stossel has not done any medical research on addiction or the dangers of opioids. He has therefore no understanding that the majority of addiction, overdoses and deaths are occurring iatrogenically...yes at the hands of our licenced doctors for inappropriately overprescribing "heroin". The FDA needs to better protect the public from Drug companies that use deceptive marketing practices and are only concerned about profit. The DEA needs to continue rooting out doctors who have created an epidemic of destruction. I do not want to see anyone suffer in pain but I do not believe that providing such dangerous and highly addictive prescription opioids is the answer. Medical research studies have shown that in the long term patients end up with pain and addiction. Until such time as a safe medication arrives on scene that does not cause addiction and serious risks, I believe the answer is the restriction of opioids. No new patients should be given opioids. In addition to education and safeguards, compassionte treatment programs must be put in place by the medical community to treat patients already taking opioids. This is a health issue not a moral issue Mr. Stossel. Check out www.banoxycontin.com and see the destruction.
Pete Jackson February 27, 2010 5:10 am (Pacific time)
Marianne thank you for responding to Mr. Stossel. He has lapped up the drug companies' propaganda like a thirsty dog. I would say thanks for straightening him out, but I imagine he is too stubborn to see the point you made so well. But the readers will hopefully benefit from your history lesson. When people think of OxyContin and Purdue Pharma, they need to think of a loaded gun left laying on a kitchen table. Yes, a child could kill themselves if they found it and started to play with it, but that doesn't mean we blame the kid and that it is OK to leave a loaded gun on the table. We would blame the parent who left it there. Same thing with potent prescription opioids like OxyContin - the statistics (which Mr. Stossel conveniently does not mention) confirm that prescribing of these dangerous drugs is skyrocketing. This suggests sever OVERprescribing, rather than underprescribing, of thie entire class of drugs. Young people continue to make bad choices - hey what's new - but then again the choices they have are way different than when I was young. That's because these heroin-like drugs are widely available, and they wrongly assume them to be safe (doctor-prescribed). Get a reality check-up, Mr. Stossel. Or at least a heart.
[Return to Top]©2026 Salem-News.com. All opinions expressed in this article are those of the author and do not necessarily reflect those of Salem-News.com.