
Publisher:
Bonnie King
CONTACT:
Newsroom@Salem-news.com
Advertising:
Adsales@Salem-news.com

~Truth~
~Justice~
~Peace~
TJP
Sep-02-2014 01:35

 TweetFollow @OregonNews
TweetFollow @OregonNews
Why Margaret Hamburg, MD should be fired as Commissioner of the FDA!
Marianne Skolek Salem-News.com Investigative ReporterThe epidemic is getting worse and still the drugs keep coming.
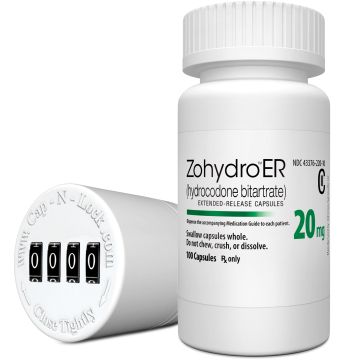 Zohydro contains up to five times the amount of narcotic hydrocodone previously available in pills. |
(MYRTLE BEACH, SC) - Some interesting events occurred this past week in the prescription opioid/heroin epidemic paralyzing the U.S. and Canada. Dr. Hamburg, Bob A. Rappaport, MD and Douglas Throckmorton, MD need to be relieved of their duties at the FDA immediately.
It is not enough to ask for the resignations of these individuals who have been accomplices in the prescription opioid/heroin deaths and addictions.
Let me lay out some facts that indicate why we are losing a generation of young people to the opioid/heroin epidemic in the U.S. and Canada while the FDA has had its eyes wide shut.
On March 1, 2013 the FDA issued the following directive entitled "Attention Prescribers":
FDA seeks your help in curtailing the U.S. opioid epidemic
FDA is asking all prescribers of opioids to ensure they have thorough knowledge of the FDA-approved product labeling for the opioids they prescribe, and to ensure they have adequate training in opioid therapy. Below is our Open Letter to prescribers. We encourage all prescribers to help curb our nation's opioid epidemic!
View and print full article1 - Attention Prescribers: Curtailing Opioid Epidemic [PDF - 137KB]
Here are some highlights of the FDA's plea to prescribers to "curtail the opioid epidemic".
In light of the expanding opioid epidemic in the U.S., FDA urges prescribers to take advantage of training on opioid prescribing, available as of March 1, 2013. This voluntary training will be provided at little to no cost through accredited continuing education activities supported by independent education grants.
Taking advantage of training opportunities on opioid therapy, now and in the future, is one of three key roles that FDA sees for prescribers in helping to curtail this pervasive problem.
The other two are: knowing the content of the most current drug labels for the opioids they prescribe, and educating patients about the appropriate use of opioids, their potential risks, and proper disposal techniques.
Misuse and abuse of prescription opioids has reached epidemic proportions in the U.S. While much of the problem is attributable to illicit use, appropriate use of medications for pain may also lead to unnecessary adverse events, addiction, and death for some patients. No group can be more effective in reducing this trend than our nation’s front-line health care professionals, especially physicians and other prescribers.
FDA seeks your help in curtailing the U.S. opioid epidemic? Isn't it the FDA who should be curtailing the US opioid epidemic? Not doing your job Drs. Hamburg, Rappaport and Throckmorton and we are losing a generation of young people because of your incompetence.
This past week six New England governors urged the U.S. Health and Human Services (HHS) secretary, Sylvia Burwell to reverse the Food and Drug Administration's approval of a powerful new painkiller, Zohydro. In their letter to Secretary Burwell, the governors of Vermont (Peter Shumlin), New Hampshire (Maggie Hassan), Maine (Paul LePage), Massachusetts (Deval Patrick), Connecticut (Dan Malloy) and Rhode Island (Lincoln Chafee) lay out that their states are in the midst of an addiction crisis.
The drug Zohydro is an extended-release capsule that contains up to five times the amount of narcotic hydrocodone previously available in pills. The letter says the drug's maker, Zogenix is preparing to seek approval for an abuse-deterrent formulation.
The governors say the likelihood of addiction and abuse of the current drug and the anticipated availability of an alternative are grounds to rescind the approval. They further indicated their states don't need another high-risk drug.
In a separate action, the Governors wrote to Canadian Health Minister Rona Ambrose and Minister of Public Safety and Emergency Preparedness Steven Blaney in support of Health Canada’s decision to require abuse deterrence for certain controlled substances. That requirement currently exists for many of these drugs sold in the United States, but the Governors have been concerned about the smuggling of medications without abuse-deterrent measures from Canada. Those measures make it difficult or impossible for the drugs to be crushed or injected, for example, and harder to abuse.
The six governors minus Connecticut's Dan Malloy are to be commended for their efforts in recruiting the Health and Human Services Secretary to call out the FDA's irresponsibility in approving a very dangerous opioid, Zohydro without any safe-guarding abuse deterrent built in. Zohydro was approved over the objections of the FDA's Advisory Committee.
My problem here is with Governor Malloy's involvement in penning a letter with five other governors when he has very tight connections financially to the maker of OxyContin, Purdue Pharma. Next week in Part 2, the Malloy OxyContin connection.
Anyone who has lost a loved one to death or addiction to prescription opioids/heroin is encouraged to come to Washington, DC on September 28, 2014 and have your voice heard at the FED UP Rally. I am honored to be a speaker at the Rally. A brochure is shown (below) with details of this event.
It is time for those who can make a difference to let the FDA and elected officials in Washington, DC know that we want to save future generations from a pharma/FDA generated prescription opioid/heroin epidemic -- and we want it now.
LP - So you know I don't like to dance because of being too lethargic, but feel free to learn the quota dance. Love you.


Marianne Skolek is an Investigative Reporter focusing on the Prescription Opioid/Heroin Epidemic in the U.S. and Canada. In particular, Marianne has covered the criminal marketing of OxyContin going back to 1999 and continuing to the present.
In 2002, Marianne lost her daughter, Jill to prescribed OxyContin which her physician referred to as "mobility in a bottle." It was, in fact, death in a bottle. After doing extensive research on the maker of OxyContin, Purdue Pharma, Marianne began working with the Department of Justice in Virginia in their criminal investigation into Purdue Pharma and in July 2007 was asked by the U.S. Attorney John Brownlee prosecuting the case to testify against the three CEO's of Purdue Pharma, Michael Friedman, Paul Goldenheim, MD and Howard Udell, Chief Counsel. The CEO's pleaded guilty to misleading the medical profession about the dangers of OxyContin. Marianne also testified against Purdue Pharma at a Judiciary Hearing of the U.S. Senate in July 2007.
Marianne's research, writing and contact with government agencies and attorneys has also exposed the heavily funded pain foundations set up by the pharmaceutical industry and their paid physician spokespersons who convinced the medical boards in 50 states and Canada that dangerous opioids such as OxyContin were less likely to be addictive. These physicians also downplayed the risks of addictive opioids in books as authors. These books are still available for sale and promoted to the medical profession.
 |
Articles for September 2, 2014 | Articles for September 3, 2014
Salem-News.com:





Terms of Service | Privacy Policy
All comments and messages are approved by people and self promotional links or unacceptable comments are denied.
Marianne Skolek September 3, 2014 10:47 am (Pacific time)
Just to clarify, the comment below credited to "Marianne" is not me. I use my first and last name when I report or comment.
Steve Gelfand, MD September 2, 2014 12:43 pm (Pacific time)
The FDA-SPONSORED REMS CME programs for ER-OPIOIDS are anything but independent and objective. Funded by the opioid industry, they provide misinformation, including the misassumption that opioids are safe and effective long-term for all types of chronic pain (as the "gold standard"), they "lump" together chronic cancer with chronic noncancer pain in which the risk/benefit ratio is high, and mislead physicians into believing they can detect and treat addiction with careful monitoring AFTER ER-opioids have been prescribed. The goal here is not to reduce opioid prescribing, but to maintain it-to the economic benefit of the opioid and pain industries.
Marianne September 2, 2014 6:33 am (Pacific time)
Kudos for your courage to speak truth to power. I hope you will also have something to say about the IPRCC developing a National Pain Strategy with same of the same professionals that fueled the opioid epidemic. It is amazing to me- given the lack of evidence for longterm benefits of opioids for noncancer pain- the Federal Government continues their poisonous enthusiasm for such dangerous medications. We could label such enthusiasm the "cash cow" anti- pattern-but whatever we label it, enthusiasm for more opioids is poor practice and unscientific.
[Return to Top]©2026 Salem-News.com. All opinions expressed in this article are those of the author and do not necessarily reflect those of Salem-News.com.